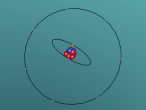
Nitrogen Atom
Nitrogen
Nitrogen is one of the many elements that make up the universe, and it is quite prominent on earth. In fact, it is present in all living organisms - including animals and plants - and makes up 78% of air. It occurs in nature as the colourless and odourless nitrogen gas (N₂), a diatomic molecule held together by a triple convalent bond, and is similar to water as a liquid. While nitrogen itself is not inert (i.e. chemically inactive), this bond allowed it to be inert enough to be used in various applications where a chemical reaction is undesired, i.e. a 'chemical blanket' which separates flammable objects from oxygen. In general, nitrogen is unreactive at normal temperatures and pressures, making it useful.
It is named from the Greek words nitron and genes, meaning 'nitre forming' due to its presence in saltpeter (which is also known as potassium nitrate; KNO₃). It was discovered by Daniel Rutherford in 1772, who removed oxygen (O₂) and carbon dioxide (CO₂) from the air and determined that the remaining gas cannot be burnt, although he did not recognise it as a different substance. In the same year, Carl W. Scheele, a French chemist, determined that it is a constituent of air, but he did not receive much recognition due to Rutherford publishing before him. Nitrogen was first considered a chemical element by the French Antonie-Laurent Lavoiser, who named it azote due to its apparent inability to support life.
Nitrogen has many applications. Nitrous oxide (N₂O), commonly known as laughing gas, is detained in surgery and dentistry to induce anaesthesia. Nitrogen is also used, along with hydrogen, in ammonia (NH₃), which is often applied in fertilisers or as a household disinfectant. It is often considered a cheaper alternative to argon when used in incandescent light bulbs. One of its more prominent usages is preservation by replacing air in packages, preventing spoilage by oxidation, mold or moisture. Liquid nitrogen boils at -196℃ and freezes at -210℃; this characteristic has led it to be used for its cold properties, furthering the extent it is used to store food and the alike.
There are many other things about nitrogen that can be further investigated, like the 'nitrogen cycle', in which the nitrogen cycles through the environment and organisms by being converted into different forms. It would also be interesting to research more on its allotropes and isotopes, along with how its compounds are discovered. The hazard it poses, despite being a useful chemical, is also of interest. For example, 'nitrogen asphyxiation' was elected to be used as a - in comparsion to other methods - humane execution, which spurred many news articles about the subject. So it seems, nitrogen is used in a wide array of applications and is involved with many aspects of life.
http://www.nature.com/scitable/knowledge/library/the-nitrogen-cycle-processes-players-and-human-15644632, http://www.visionlearning.com/en/library/Earth-Science/6/The-Nitrogen-Cycle/98
http://environmentalchemistry.com/yogi/periodic/N-compounds.html
http://www.ch.ic.ac.uk/widdowson/teach_files/nitrogen/dw1.html
http://www.monash.edu/ohs/information-and-documents/all-information-sheets/handling-and-storage-of-liquid-nitrogen
News Articles:
http://www.slate.com/articles/news_and_politics/jurisprudence/2014/05/death_by_nitrogen_gas_will_the_new_method_of_execution_save_the_death_penalty.html
http://www.politico.com/magazine/story/2015/04/oklahoma-death-penalty-gas-chamber-117156
http://www.spiegel.de/international/world/us-representative-wants-to-replace-lethal-injection-with-nitrogen-a-991651.html
http://time.com/3749879/nitrogen-gas-execution-oklahoma-lethal-injection/
http://www.npr.org/sections/thetwo-way/2015/04/17/400444876/oklahoma-approves-nitrogen-asphyxiation-for-executions
It was quite difficult to create the atom in the VRMath 2.0 program. Although the basic idea of the atom was captured, it was unable to be animated due to the difficultly of aligning the various objects together. The usage of mathematics is quite interesting, and allowed for more precision during the modelling process, although it is also time-consuming to correctly calcuate proportions and the alike. Eventually the electronic tool utilised - at first an iPad - had been replaced with a laptop, which allowed for easier use and less minor incidents, which ranged from crashing the webpage to grossly disfiguring the in-progress atom. Most of the time spent coding is experimenting with various values and commands - which piled up into a large amount of time - but in the end, it is an interesting experience.
Futher reading:
http://www.uigi.com/nitrogen.html
https://www.britannica.com/science/nitrogen
http://chemwiki.ucdavis.edu/Core/Inorganic_Chemistry/Descriptive_Chemistry/Elements_Organized_by_Block/2_p-Block_Elements/Group_15%3A_The_Nitrogen_Family/Chemistry_of_Nitrogen
VRMath 2.0 Program:
https://vrmath2.net/VRM2/
Groups: