Fluorine Atom
Fluorine Blog
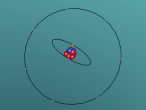
The atom I have chosen is Fluorine (F). Fluorine is major part of everyday lives. For example, fluorine is found in toothpaste, which is an essential item used on a daily basis. Ever wondered why some lights are white? This is because they are fluorescent lights, made from fluorine. Fluorine is one of many elements and follows the same structure; protons and neutrons found in the nucleus, with electrons on the outer shell. Its atomic number is 9 and its mass is 19. Fluorine is the first element in group 17 (the Halogen group). (LibreTexts, 2015).
Composition
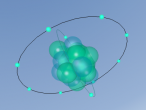
Within the atom, a nucleus is present in the middle, surrounded by shell/s of electrons. The nucleus contains protons, in which are positively charged. The number of protons in the nucleus is the same number of negatively charged electrons positioned on the shell/s. The nucleus also contains neutrons, with no charge. (No author/date). In terms of the atom Fluorine, it contains 9 protons and 10 neutrons within the nucleus. Therefore, it also contains 9 electrons.
Structure
All atoms have shells. These shells contain a certain amount of electrons. Each atom is different in its number of electrons. The fluorine atom has nine electrons. Two of these are located on the first shell (inner shell) while the remaining seven fit along the second shell (outer shell). (Bitesize, 2014). Furthermore, fluorine is unique with its 9 electrons while other atoms have different numbers of electrons.
Links
For more information on the characteristics of fluorine, visit these websites:
https://khanacademy.org/science/chemistry/atomic-structure-and-properties
https://khanacademy.org/science/biology/chemistry--of-life/electron-shells-and-orbitals/a/the-periodic-table-electron-shells-and-orbitals-article
Some further questions into fluorine:
1. What are some compounds that involve Fluorine?
2. How fast do the shells spin?
3. How fast do the electrons orbit?
4. What is the distance between each shell?
Interesting ideas in Programming
While creating my 3D model of a Fluorine atom, I encountered some interesting ideas. Such ideas as, making the shells tilt and rotate at the same time. This would add a very realistic effect to the atom. Another interesting thought is to make the nucleus clear and create small balls as the protons and neutrons. This way you can see the little protons and neutrons which then again, adds a realistic effect. These are some interesting ideas that I would enjoy to investigate further.
Links for Programming
If you wish to learn the basics of programming, visit this link:
https://vrmath2.net
Reference List
(No author/date) Retrieved July 28 2016 from,
http://www.vanderbilt.edu/AnS/Chemistry/courses/chem104/experiment1/composition/composition.htm
LibreTexts (2015) Retrieved July 28 from,
http://chem.libretexts.org/Core/Inorganic_Chemistry/Descriptive_Chemistry/Elements_Organized_by_Block/2_p-Block_Elements/Group_17%3A_The_Halogens/Chemistry_of_Fluorine
Bitesize (2014) Retrieved July 28 from,
http://www.bbc.co.uk/schools/gcsebitesize/science/add_aqa_pre_2011/atomic/atomstrucrev4.shtml
Groups:











Comments
More work on structure was
More work on structure was needed. Fluorine is diatomic. What does that mean?